ClinicalTrials
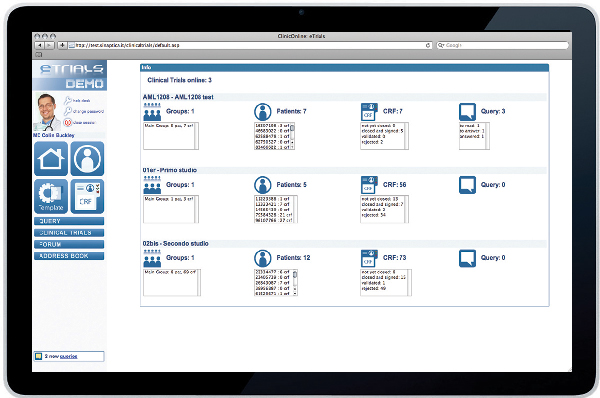
ClinicalTrials is the web based Sinaptica IT platform aimed at clinical trials data capture. Thanks to ClinicalTrials clinical research can benefit from a dedicated highly performing IT tool, efficient and cost-effective, that guarantees secure data transfer meeting the needs of any kind of clinical study.
A web based access allows you to have data available online in real time streamlining workflow, data control and analysis. ClinicalTrials can be used with any web browser (Internet Explorer, FireFox, Safari, Opera) without installing any further software and without any hardware or operative system constraint.
ClinicalTrials characteristics make it a perfect tool within an international clinical trial environment:
- Bilingual version available (English - Italian).
- A web based platform for a worldwide access, as long as an Internet connection is available.
- It does not need to be installed: it is possible to activate centers and users in remote mode.
ClinicalTrials is a tool that offers full control and whole flexibility to manage clinical trials data. Thanks to its parametric structure it can be adapted to the needs of any clinical trial protocol. Indeed, you can generate and modify, completely on your own, eCRFs (electronic Case Report Forms) and its workflow, just as access profiles can be personalized according to the needs of any of the different actors playing a role in the study (Data Manager, Biostatistician, Investigator, System Administrator and Patient).
ClinicalTrials uses the HTTPS web data transfer protocol through the SSL code system at 128 bits that guarantees maximum data security and a security logic fully compliant with Italian Data Privacy regulations (196/2003); furthermore, it has been designed to adhere to GCP (Good Clinical Practice) and to specific requirements concerning Data Privacy within the clinical research field (24 July 2008 - Italian Data Protection Supervisor Guidelines).
![]() Download ClinicalTrials Presentation (2,0 MB)
Download ClinicalTrials Presentation (2,0 MB)
![]() Download ClinicalTrials User's Guide for Investigators (8,6 MB)
Download ClinicalTrials User's Guide for Investigators (8,6 MB)


